Where does energy come from unit vocabulary -
Photosynthesis: the process by which green plants and some other organisms use sunlight to synthesize foods from carbon dioxide and water
Chloroplast: a cell organelle that contains chlorophyll and in which photosynthesis takes place.
Eukaryotic: An organism whose cells contain a nucleus surrounded by a membrane and whose DNA is bound together by proteins
Prokaryotic: a unicellular organism that lacks a membrane-bound nucleus
Oxidize: combine or become combined chemically with oxygen.
Cellular respiration: The chemical process that generates most of the energy in the cell, supplying molecules needed to make the metabolic reactions
Mitochondria: an organelle found in most cells; where cellular respiration and energy production occur.
Food pyramid: a nutritional diagram in the shape of a pyramid
Carbohydrate: a nutrient that provides energy, typically made of sugars or starches
Protein: a nutrient that is especially for the growth and repair of body tissue; building blocks
Lipids (fat): The body uses fat as a fuel source, and fat is the major storage form of energy in the body.
Consumption: the using up of a resource, often used in the context of eating
Food: any nutritious substance that people or animals eat or drink, or that plants absorb, in order to maintain life and growth.
Glucose: a simple carbohydrate (sugar) that is an important energy source in living organisms and is a component of many carbohydrates
ATP: Adenosine triphosphate; a compound that releases energy during the process of cellular respiration
Basal Metabolic Rate: the rate at which the body uses energy while at rest to keep vital functions going, such as breathing and keeping warm
Calories: a unit used in measuring the amount of energy food provides when eaten and digested:
Metabolism: the chemical processes that occur within a living organism in order to maintain life
Circulatory System: the body system that circulates blood through the body, consisting of the heart, blood vessels, and blood
Ecology and Populations (words for TIP chart and test on Tuesday May 9)
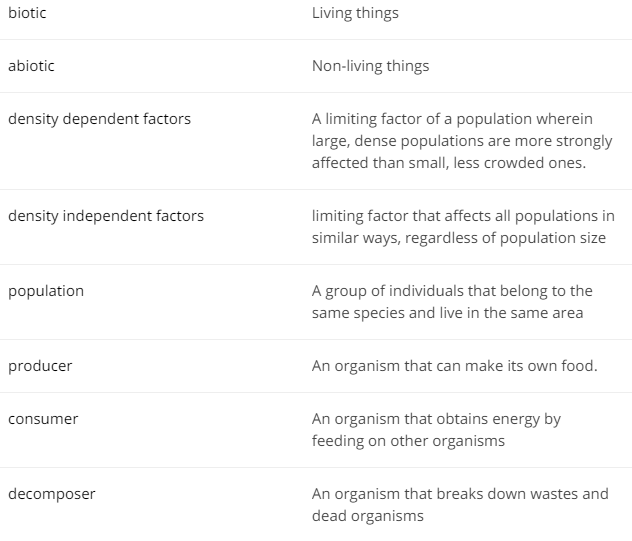
Biotic Factor - A living feature of an ecosystem (plants, animals, leaves, etc.)
Abiotic Factor - A non-living feature of an ecosystem (rock, water, sun, etc.)
Biodiversity - the variety of life in the world or in a particular ecosystem or habitat
Density-dependent factor - The tendency for the death rate in a population to increase, or the birth or growth rate to decrease, as the density of the population increases
Density-independent factor - The tendency for the death, birth or growth rate in a population neither to rise nor fall as the density increases
Population - a group of individuals of one species in an area
Producer - an organism that can make its own food
Consumer - an organism that obtains energy from feeding on another organism
Decomposer - organisms that break down waste and the remains of other organisms to return the raw materials back to the ecosystem
Coexistence - the living together of two organisms or species in the same habitat without trying to eliminate the other
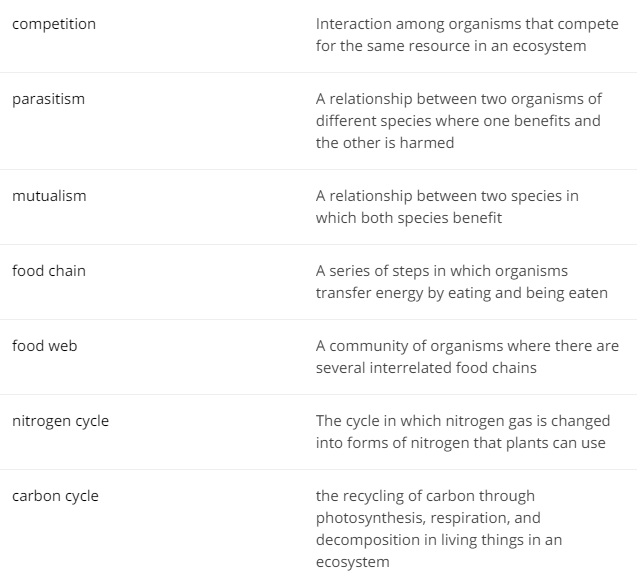
Competition - an interaction between organisms or species for resources - food, shelter, etc.
Parasitism - a relationship between two organisms in which one benefits or lives off the other
Mutualism - a relationship between two species of organisms that both benefit from the relationship
Food Chain - the order in which matter and energy in the form of food is transferred from one organism to another
Food Web - a network of food chains or feeding relationships
Nitrogen Cycle - a continuous series of natural processes by which nitrogen passes successively from air to soil to organisms and back to air or soil
Carbon Cycle - the transfer of carbon through naturally occurring processes between organisms and the environment
Water Cycle - the continuous movement of water on, above, and below the Earth’s surface
Lifeforms
Adaptation: a trait that increases the chances that an organism will survive and reproduce
Artificial selection: the breeding of plants and animals with desired traits to attempt to produce offspring with these same traits.
Biological evolution: the change over time of living organisms
Darwin: was a naturalist who proposed and provided scientific evidence that all species of life have evolved over time from common ancestors through the process he called natural selection.
Evolution: the change in population of a species over time.
Extinction: The evolutionary termination of a species caused by the failure to reproduce and the death of all remaining members of the species; the natural failure to adapt to environmental change.
Mutation: a random change to a gene that results in a new trait
Natural selection: survival of the fittest organisms that are the best adapted to their environment and the ones that will live long enough to reproduce and pass on their favorable adaptations
Species: the most specific classification of living things
Speciation: the process of natural selection producing a new species out of existing species over thousands or millions of years.
Theory: an explanation that ties together many hypothesis and observations.
Landforms
Fossil - the remains or impression of a prehistoric organism preserved in petrified form or as a mold or cast in rock
Index Fossil - a fossil that is useful for dating and correlating the strata in which it is found
Paleontologist - scientist that studies fossils
Relative Age - the geologic age of a fossil organism, rock, or geologic feature or event defined relative to other organisms, rocks, or features or events rather than in terms of years
Law of Superposition - a basic law of geology, stating that in any undisturbed sequence of rocks deposited in layers, the youngest layer is on top and the oldest on bottom, each layer being younger than the one beneath it and older than the one above it
Fault - a fracture or fissure in the Earth's crust in which there is movement on either side of the fracture; a place where sections of the crust of the Earth move relative to each other
Continental Drift - a theory set forth in 1912 by Alfred Wegener, a geophysicist and meteorologist, that explained how continents shift position on Earth's surface and why look-alike animal and plant fossils, and similar rock formations, are found on different continents
Plate Tectonics - the two sub-layers of the earth's crust (lithosphere) that move, float, and sometimes fracture and whose interaction causes continental drift, earthquakes, volcanoes, mountains, and oceanic trenches
Pangea - - a hypothetical supercontinent that included all current land masses, believed to have been in existence before the continents broke apart during the Triassic and Jurassic Periods
Evolution - any process of formation or growth; development
Geologic Time Scale - the succession of eras, periods, and epochs as considered in historical geology; the period of time covering the formation and development of the Earth, from about 4.6 billion years ago to today
Absolute Age - the true age of a rock or fossil, tells scientists the number of years ago a rock layer formed
Natural Resources
Renewable: energy from a source that is not depleted when used
Nonrenewable: an energy source that can not be replaced by natural means at the same rate that it is consumed.
Conservation: preservation, protection, or restoration of the natural environment, natural ecosystems, vegetation, and wildlife.
Depeletion: reduction in the number or quantity of something.
Fossil Fuels: a natural fuel formed in the past from the remains of living things. (ex: coal, oil, natural gas)
Hydropower or Hydroelectric Energy: energy that comes from the force of moving water.
Nuclear Energy: energy stored in the nucleus of an atom.
Biomass: organic matter that can be used as an energy source. (ex: wood, crops, and animal waste)
Wind Energy: energy harnessed from air in motion.
Solar Energy: energy radiated from the sun.
Solar Cell: device converting solar radiation into electricity
Geothermal Energy: heat energy from within the earth.
Recycling: making waste usable again or repurposing (using for new purpose)
Biotechnology Vocabulary
Biotechnology: the exploitation of biological processes for industrial and other purposes, especially the genetic manipulation of microorganisms for the production of antibiotics, hormones, etc.
Biofuels: A type of energy derived from renewable plant and animal materials or organic matter.
Bioremediation: introduced microorganisms that break down environmental pollutants, in order to clean up a polluted site
Cloning: propagate (an organism or cell) to make an identical copy of
Genetic Engineering: DNA manipulation of cells or organisms- Genes are added, deleted or changed. Genetic Modification: allows selected individual genes to be transferred from one organism into another and also between non-related species. (GMO)
Mutagen: a toxic chemical that causes damage to an organism’s DNA.
Mutation: a random change to a gene that results in a new trait.
Regeneration: the action or process of regrowth; in particular the formation of new animal or plant tissue. Stem Cells: a simple cell in the body that is able to develop into any one of various kinds of cells(such as blood cells, skin cells, etc.)
Disease Unit - Set 1
Disease - a change that disrupts normal function of a body system.
Infectious Disease - a disease caused by a pathogen that can be spread (that is communicable or contagious)
Noninfectious Disease - a disease that can not be spread (you can't catch it)
Microbe - an organism or pathogen that can not be seen with just your eye (you need an microscope)
Pathogen - a microbe that causes disease.
Bacteria - a unicellular organism (single cell)
Virus - a nonliving particle consisting of DNA encased in a protein coat that takes over a cell to replicate.
Fungus (Fungi)- a multicellular (eukaryotic) organism including mold, yeasts, and mushrooms.
Parasite - an organism that lives in or on another organism (its host)
Disease Set 2
Carrier - a person that passes along a disease that they "carry" but may or may not show symptoms
Vector - an animal that 'carries' and transmits a disease
Antibiotic - medicine that kills or slows the growth of bacteria
Antibody - a chemical the body produces that destroys an invading pathogen.
Antimicrobial - a substance designed to kill microbes
Vaccine - a preventative treatment (often in the form of a shot) created to boost immunity to a specific disease - either a weakened form of the pathogen to stimulate antibody production in the body.
Epidemic - a disease that is spread across large populations of people in a single region.
Pandemic - an epidemic that spreads globally or across a large geological area.
Outbreak - a sudden start of a disease
Chemistry unit Set 5 -
Chemical Change = Chemical Reaction
Chemical Bond - sharing of an electron to make a molecule or compound
Chemical Equation - written expression of a chemical reaction
Subscript - small number that tells how many atoms there are of the element in front of it.
Coefficient - large number in front of a compound telling how many molecules there are
Combustion Reaction - a burning of a hydrocarbon (fuel) that requires O2 as a reactant and produces CO2 and H2O
Synthesis Reaction - a single new product formed from 2 or more reactants
Decomposition Reaction - opposite of a synthesis - a single reactant breaks down or decomposes to create 2 or more products
Single Replacement Reaction - a reactant switches element it is bound to to create a new compound (one reactant does not have a partner)
Double Replacement Reaction - both reactants switch partners to create 2 new products.
Chemistry Subunit 4 -
Chemical Change - a chemical reaction that is permanent with indicators such as change in color, temperature, and odor, fizzing or foaming, creation of light, formation of a precipitate .
Physical Change - a change that is easily reversible.
Appearance - a physical trait
Exothermic -chemical reaction giving off heat.
Endothermic - chemical reaction absorbing or taking in heat.
Reactant - what goes into a chemical reaction (on the left side of a chemical equation).
Product - what comes out of a chemical reaction (on the right side of the chemical equation)
Reaction - a chemical change
Solution - a homogeneous mixture where a liquid solvent (typically water) dissolves a solute (a solid) in a uniform concentration. THINK SUGAR IN YOUR TEA!
Precipitate - white solid that is the product of a chemical reaction when two aqueous solutions are mixed.
Chemical reaction - chemical change - rearrangement of elements to form a new compound.
Law of Conservation of Mass - law that states matter is neither created nor destroyed.
catalyst - a substance that starts or speeds up a chemical reaction (movement, heat, or a chemical)
Chemistry Subunit 3 Vocabulary
Metal- a solid material that is typically hard, shiny, malleable, fusible, and ductile, with good electrical and thermal conductivity
Metalloid- an element (e.g., germanium or silicon) whose properties are intermediate between those of metals and solid nonmetals. They are electrical semiconductors.
Nonmetal- a chemical element (as boron, carbon, or nitrogen) that lacks the characteristics of a metal and that is able to form anions, acidic oxides, acids, and stable compounds with hydrogen.
Chemical Properties - things you can not see - like solubility,
Physical Properties - an attribute you can see; appearance
Reactivity - degree to which something reacts
Solubility - ability to go into solution (a chemical property)
Density - how close particles are; compactness; mass per unit volume
Boiling Point - temperature that a liquid changes to a gas
Melting Point - temperature that a solid changes to a liquid
Periodic Table Family - groups or columns on PT that have similar chemical properties
Bohr Model - depicts or shows the electron configuration (number and location of electrons)
malleable - pliable or shapeable
ductile - can be pounded into a thin wire
Chemistry Subunit 2 - vocabulary test on 11/10
Chemistry Vocabulary Unit 2
Homogeneous - equal concentrations or distribution throughout.
Heterogeneous - uneven distributions or concentrations throughout
Periodic Table- A table of elements, arranged by atomic number that shows the patterns in their properties.
Subatomic Particles- a particle smaller than an atom
Protons- a stable subatomic particle occurring in all atomic nuclei, with a positive electric charge
Electrons- subatomic particle with a negative charge located in the electron cloud
Neutrons- A particle in the nucleus of an atom that has no charge.
Atomic mass - The number of both protons and neutrons in the nucleus.
Atomic number- The number of protons in the nucleus of an atom.
Chemistry - Matter Subunit- 1
Matter - something that occupies space, has mass, and can exist in liquic, gas, or solid, form.
Atoms-The smallest part of an element. A group of the same atoms together are called an element.
molecule - more than one atom chemically bonded together
Element - all the same type of atoms make up elements (currently over a 120 on periodic table) - it can not be broken down into smaller units.
Compound - a substance made up of two or more substances joined by chemical bonds.
Mixture - two or more substances not chemically combined (lemonade).
Solution - A solution is a mixture where one of the substances dissolves in the other. The substance that dissolves is called the solute. The substance that does not dissolve is called the solvent. An example of a solution is salt water.
Fresh water - set 2
Clean water act - 1972 - Enacted by the USEPA to regulate water pollution
Safe Water Drinking - 1974 - Enacted by USEPA to ensure drinking water is maintained at safe levels for public
DENR - Department of Environment and Natural Resources. This is a North Carolina state agency designed to enforce laws pertaining to the environment.
USEPA - 1970 - federal agency established to protect human health and the environment.
Point source - a single definable source of pollution
Nonpoint source - pollution from diffuse or multiple undefined sources.
Phosphate - a salt form of phosphorus that is found in fertilizer
Nitrate / Nitrite - forms of nitrogen that are found in fertilizer
Algal Bloom - excess algae growth as a result of eutrophication
Eutrophication - excess nutrients typically from excess runoff resulting in an algal bloom which results in aquatic organism death due to a lack of oxygen.
Turbidity - a measure of total suspended solids or clarity of the water
Not these yet but see fresh water set 1 list below
Organic Material - substances in the soil that were once alive
Inorganic Material - sythetic material or not organic material
Water Reclamation - waste water treatment plant - the process of making water safe for discharge into the environment
Water Treatment - process of making water potable or drinkable
Clarification - process of treating water and making it clear.
Sedimentation - process of treating water and allowing larger particles to settle out to the bottom.
Coagulation - the process of treating water that cause particles to clump together
Fresh water set 1
pH - a measure of acidity or basicidity ranging from 0 - 14 with the lower the number being more acidic and the higher the number the more basic.
D.O. - dissolved oxygen - the amount of oxygen in the water. The range being from 0 - 18 with 9 being saturation point at standard temperature and pressure.
Stewardship - the careful and responsible management of something entrusted to you (like the environment).
Watershed - the land determining the drainage direction of water (typically by altitude).
Bioindicator - a living organism used to determine the quality of water (macroinvertebrate).
Tributaries - a moving body of water in size between a creek and a river
There are 4 sets of vocabulary on the first test.
Oceanography Vocab set 2
Salt Marshes - type of wetland in cooler areas further from equator rich in nutrients, grasses and habitat hold soil in place
Mangrove Forests - a type of wetland found in tropical regions. Roots protect shoreline.
Coral Reefs - limestone build up of microscopic organisms that eat algae and are home to 25% of all ocean life. Coral reefs are endangered due to pollution and increased ocean temperatures from global warming.
Kelp forest - sea weed that can be up to 40 feet deep that float to the surface by air bulbs. They are a habitat for fish and marine animals.
wetlands - swampy, marshy lands at the edges of estuaries
erosion - gradual wearing away of land from wind or water
nutrients - substance that provides nourishment essential for growth and the maintenance of life.
extremophiles - organisms living in deep water under extreme temperature, pressure, pH, or chemical conditions.
bioluminescence - light coming from a living organism
coreolis effect - the effect tends to deflect moving objects to the right in the northern hemisphere and to the left in the southern and is important in the formation of cyclonic weather systems.
currents - movement of water due to temperature or density
tides - movement of water due to lunar attraction.
thermohaline - large-scale ocean circulation that is driven by global density gradients created by surface heat and freshwater fluxes.
thermocline - mixing zone of cold and warm water
invasive species - a nonnative organism that threatens the ecosystem when introduced into a new habitat.
Quiz on these hydrology vocabulary on 9/16
1.) hydrology - study of water
2.) hydrosphere - all the waters on the earth's surface, such as lakes and seas, and sometimes including water over the earth's surface, such as clouds.
3.) aquifer - a body of permeable rock that can contain or transmit groundwater.
4.) water cycle - a closed continuous circuit of water - including condensation, infiltration, evaporation, run-off, frozen water, precipitation, and transporation
5.) groundwater - water under the surface of the earth
6.) river basin - the area of land that a river and tributaries drain into
7.) contaminants - anything polluting the water
8.) Ocean (salt water) - five continuous saltwater bodies making up 71 % of the earths surface
9.) Marine - having to do with the ocean or sea
10.) Biosphere - the regions of the surface, atmosphere, and hydrosphere of the earth (or analogous parts of other planets) occupied by living organisms.
11.) Lithosphere - the rigid outer part of the earth, consisting of the crust and upper mantle.
12.) Atmosphere - the envelope of gases surrounding the earth or another planet.
13.) Freshwater - water that does not contain salt
Week 3 words
upwelling - a rising of seawater, magma, or other liquid brining nutrients from the bottom of the ocean.
downwelling - the downward movement of fluid bringing oxygen from the sunlight zone
hydrothermal vents - an opening in the sea floor out of which heated mineral-rich water flows
oceanography - the study of the ocean
sunlight zone (euphotic or phototrophic) - the depth of the ocean where photosynthesis occurs.
twilight zone (disphotic) - the lowest level of the ocean to which light can penetrate but where there is limited or no photosynthesis.
intertidal zone - the area that is above water at low tide and under water at high tide (in other words, the area between tide marks)
abyssal zone - At depths of 4,000 to 6,000 metres (13,123 to 19,685 feet), this zone remains in perpetual darkness and never receives daylight.
plankton - microscopic organisms drifting or floating in the sea or fresh water, consisting chiefly of diatoms, protozoans, small crustaceans, and the eggs and larval stages of larger animals. Many animals are adapted to feed on plankton, especially by filtering the water.
salinity - a measure of salt content in something. In the ocean the salinity is typically 35 - 36 parts per thousand.
brackish water - a mixture of salt and fresh water.
estuary - the tidal mouth of a large river, where the tide meets the stream.
quiz 9/9 on these words -Vocabulary set 1 - Water Properties Vocabulary
Absorbency: The ability to take in a material.
Capillary Action: The process that moves water through a narrow porous space. Adhesion: The tendency of water particles to stick to other surfaces. Cohesion: The tendency of water molecules to stick together. (Attractive force between water molecules)
Surface Tension: The force that acts on the particles at the surface of a material to hold them together.
Buoyancy: The ability of a fluid to exert an upward force on an object that is immersed in it.
Polarity: Uneven sharing of electrons across a molecule.
Universal Solvent: The quality of water that makes it able to dissolve more substances that any other solvent.
Heat of Vaporization: The amount of heat required to change a substance from a liquid to a gas.
Specific Heat: The amount of heat needed to raise the temperature of something 1 degree Celsius
solute: substance that is dissolved into solution
solvent: liquid that dissolves something
Vocabulary 9 Chemical Reactions
Chemical Bond - sharing of valence electrons between elements
Chemical Reaction - rearrangement of elements to form a new compound
Law of Conservation of Matter - matter is neither created or destroy
Product - what comes out of a chemical reaction
Reactant - what goes into a chemical reaction
catalyst - physical or chemical that starts a chemical reaction -
(movement, heat, or chemical)
subscript - small number that tells how many of an element are
in a compound - the 2 in H20
coefficient - number in front of the element that tells how many
of a compound are present.
synthesis reaction - 2 compounds combine to form a new product.
decomposition reaction - a compound breaks down into 2
or more components
combustion reaction - a chemical reaction where O2 must be a reactant
single replacement reaction - replacing one element for another
double replacement reaction - trade partners in a chemical reaction
Vocabulary 8 Physical and Chemical Change
Appearance - physical way something looks -dull, shiny, luster, rough, soft, etc
Boiling point - temp that something changes from liquid to gas
Chemical Properties - property only observed when there is a change of the substance identity
Physical Change - changing the state or appearance of a substance
Chemical Change - change in chemical composition due to a reaction
Physical Properties - appearance, density, measurable, whose value describes a state of a physical system
Bohr model - model showing the electron configuration of an element
Law of Conservation of Mass - states that mass in an isolated system is neither created nor destroyed by chemical reactions or physical transformations. According to the law of conservation of mass, the mass of the products in a chemical reaction must equal themass of the reactants.
Reactants - substances that interact or react.
Reactivity - a measure of how two substances will intereact
Precipitate - solid formed from the reaction of two liquids
Vocabulary 7 - Atoms and Periodic Table Vocabulary Quiz 11/6
Periodic Table- A table of elements, arranged by atomic number that shows the patterns in their properties.
Subatomic Particles- a particle smaller than an atom
Protons- a stable subatomic particle occurring in all atomic nuclei, with a positive electric charge
Electrons- subatomic particle with a negative charge located in the electron cloud
Neutrons- A particle in the nucleus of an atom that has no charge.
Atomic mass - The number of both protons and neutrons in the nucleus.
Atomic number- The number of protons in the nucleus of an atom.
Metal-An element that tends to be shiny, easily shaped, and a good conductor of heat and electricity.
Metalloid- an element whose properties are intermediate between those of metals and solid nonmetals.
Nonmetal- An element that has properties opposite of those of a metal.
Vocabulary 6 - Matter Quiz 10/30
Matter - something that occupies space, has mass, and can exist in liquic, gas, or solid, form.
Mixture - two or more substances not chemically combined (lemonade).
Exothermic -chemical reaction giving off heat.
Endothermic - chemical reaction absorbing or taking in heat.
Homogeneous - equal concentrations or distribution throughout.
Heterogeneous - uneven distributions or concentrations throughout the same
Atom - smallest building block of matter
Element - all the same type of atoms make up elements (currently over a 100)
Compound - a substance made up of two or more substances joined by chemical bonds.
Solution - a mixture in which one or more substances are uniformly distributed.
Solute - a substance that dissolves in a solvent (sugar)
Solvent - a substance that dissolves other substances. Water is the universal solvent.
Vocabulary 5 - Ocean
Upwelling- Movement of water up to the surface. Brings nutrients from the bottom of the ocean upward.
Downwelling- Movement of water downward. Brings oxygen to the depths of the ocean.
Hydrothermal Vents: An area of volcanic activity where the tectonic plates are moving apart and super-hot fluid comes through.
Oceanography- The study and exploration of the world’s oceans.
Water Reclamation - water that has been reclaimed from municipal wastewater, or sewage.
Sunlight zone (euphotic or phototropic zone) enough light penetrating the water to support photosynthesis.
Twilight zone (disphotic zone) very small amounts of light, plants do not grow here
Midnight zone (aphotic zone) without light; near freezing temperatures; very few living orgamisms; high pressure
Plankton- the small and microscopic organisms drifting or floating in the sea or fresh water (phytoplankton are plants and zooplankton are animals)
Intertidal zone-the area that is above water at low tide and under water at high tide
Vocabulary Set 4 - Fresh water
River Basin - the land that drains into a river
Tributaries - the smaller streams and creeks that build to the river
Groundwater - water found underground in aquifers
Watershed - the ridge that separates rivers draining one direction
Fresh water - waters without salt
Estuary - habitat where fresh and salt mix (rivers meet ocean)
Stewardship - taking care of the environment
Brackish water - salty and fresh water mix (not at the salinity of the ocean)
Photosynthesis - process of plants making sugars from the sun
Point Source - pollution that comes from a specific location
Nonpoint source - where there not a specific origin of the pollution.
Chemical Bond - sharing of valence electrons between elements
Chemical Reaction - rearrangement of elements to form a new compound
Law of Conservation of Matter - matter is neither created or destroy
Product - what comes out of a chemical reaction
Reactant - what goes into a chemical reaction
catalyst - physical or chemical that starts a chemical reaction -
(movement, heat, or chemical)
subscript - small number that tells how many of an element are
in a compound - the 2 in H20
coefficient - number in front of the element that tells how many
of a compound are present.
synthesis reaction - 2 compounds combine to form a new product.
decomposition reaction - a compound breaks down into 2
or more components
combustion reaction - a chemical reaction where O2 must be a reactant
single replacement reaction - replacing one element for another
double replacement reaction - trade partners in a chemical reaction
Vocabulary 8 Physical and Chemical Change
Appearance - physical way something looks -dull, shiny, luster, rough, soft, etc
Boiling point - temp that something changes from liquid to gas
Chemical Properties - property only observed when there is a change of the substance identity
Physical Change - changing the state or appearance of a substance
Chemical Change - change in chemical composition due to a reaction
Physical Properties - appearance, density, measurable, whose value describes a state of a physical system
Bohr model - model showing the electron configuration of an element
Law of Conservation of Mass - states that mass in an isolated system is neither created nor destroyed by chemical reactions or physical transformations. According to the law of conservation of mass, the mass of the products in a chemical reaction must equal themass of the reactants.
Reactants - substances that interact or react.
Reactivity - a measure of how two substances will intereact
Precipitate - solid formed from the reaction of two liquids
Vocabulary 7 - Atoms and Periodic Table Vocabulary Quiz 11/6
Periodic Table- A table of elements, arranged by atomic number that shows the patterns in their properties.
Subatomic Particles- a particle smaller than an atom
Protons- a stable subatomic particle occurring in all atomic nuclei, with a positive electric charge
Electrons- subatomic particle with a negative charge located in the electron cloud
Neutrons- A particle in the nucleus of an atom that has no charge.
Atomic mass - The number of both protons and neutrons in the nucleus.
Atomic number- The number of protons in the nucleus of an atom.
Metal-An element that tends to be shiny, easily shaped, and a good conductor of heat and electricity.
Metalloid- an element whose properties are intermediate between those of metals and solid nonmetals.
Nonmetal- An element that has properties opposite of those of a metal.
Vocabulary 6 - Matter Quiz 10/30
Matter - something that occupies space, has mass, and can exist in liquic, gas, or solid, form.
Mixture - two or more substances not chemically combined (lemonade).
Exothermic -chemical reaction giving off heat.
Endothermic - chemical reaction absorbing or taking in heat.
Homogeneous - equal concentrations or distribution throughout.
Heterogeneous - uneven distributions or concentrations throughout the same
Atom - smallest building block of matter
Element - all the same type of atoms make up elements (currently over a 100)
Compound - a substance made up of two or more substances joined by chemical bonds.
Solution - a mixture in which one or more substances are uniformly distributed.
Solute - a substance that dissolves in a solvent (sugar)
Solvent - a substance that dissolves other substances. Water is the universal solvent.
Vocabulary 5 - Ocean
Upwelling- Movement of water up to the surface. Brings nutrients from the bottom of the ocean upward.
Downwelling- Movement of water downward. Brings oxygen to the depths of the ocean.
Hydrothermal Vents: An area of volcanic activity where the tectonic plates are moving apart and super-hot fluid comes through.
Oceanography- The study and exploration of the world’s oceans.
Water Reclamation - water that has been reclaimed from municipal wastewater, or sewage.
Sunlight zone (euphotic or phototropic zone) enough light penetrating the water to support photosynthesis.
Twilight zone (disphotic zone) very small amounts of light, plants do not grow here
Midnight zone (aphotic zone) without light; near freezing temperatures; very few living orgamisms; high pressure
Plankton- the small and microscopic organisms drifting or floating in the sea or fresh water (phytoplankton are plants and zooplankton are animals)
Intertidal zone-the area that is above water at low tide and under water at high tide
Vocabulary Set 4 - Fresh water
River Basin - the land that drains into a river
Tributaries - the smaller streams and creeks that build to the river
Groundwater - water found underground in aquifers
Watershed - the ridge that separates rivers draining one direction
Fresh water - waters without salt
Estuary - habitat where fresh and salt mix (rivers meet ocean)
Stewardship - taking care of the environment
Brackish water - salty and fresh water mix (not at the salinity of the ocean)
Photosynthesis - process of plants making sugars from the sun
Point Source - pollution that comes from a specific location
Nonpoint source - where there not a specific origin of the pollution.
Vocabulary Set 3 Water Quality background words (you should know these)
Aquifer - layer of rock and soil that is full of water.
Groundwater - water that is under the ground
River Basin - the area of land that a river and tributaries drain into
Contaminants - pollutants and particulates that are in the water
Salinity - salt in the water
Upwelling - a turnover in the ocean
Density - how close together the particles are
Marine - relating to the ocean or sea
Vocabulary 3 – Water quality (you need to learn these)
pH - a measure of aciditity and basicidity - potential Hydrogen
Nitrates - a compound of nitrogen and oxygen used in fertilizer
Phosphates - a compound with phosphorus used in fertilizer
Algae blooms - excess algae growth
Water quality - determined by 6 measurement - a measurement of how clean the water is
Stewardship - taking care of the environment
Salinity - how much salt is in the water
Dissolved oxygen - how much oxygen is in the water
Eutrophication - excess nutrients in the water resulting in algae growth depleting dissolved oxygen
Acid(ic) low pH - like soda or juice or ammonia
Base(ic) high pH - like bleach
Turbidity - a measure of how many particulates are in the water - measured by how much light can pass through
Bioindicators (macroinvertebrate) - an organism that is sensitive to water quality.
Sedimentation- particles settling out of water.
Organic materials - material that decays that was once alive
Inorganic materials - materials that break down but were never alive like alluminum cans,
Vocabulary set 2 water cycle
Aquifer - layer of rock and soil that is full of water.
Groundwater - water that is under the ground
River Basin - the area of land that a river and tributaries drain into
Contaminants - pollutants and particulates that are in the water
Salinity - salt in the water
Upwelling - a turnover in the ocean
Density - how close together the particles are
Marine - relating to the ocean or sea
Vocabulary 3 – Water quality (you need to learn these)
pH - a measure of aciditity and basicidity - potential Hydrogen
Nitrates - a compound of nitrogen and oxygen used in fertilizer
Phosphates - a compound with phosphorus used in fertilizer
Algae blooms - excess algae growth
Water quality - determined by 6 measurement - a measurement of how clean the water is
Stewardship - taking care of the environment
Salinity - how much salt is in the water
Dissolved oxygen - how much oxygen is in the water
Eutrophication - excess nutrients in the water resulting in algae growth depleting dissolved oxygen
Acid(ic) low pH - like soda or juice or ammonia
Base(ic) high pH - like bleach
Turbidity - a measure of how many particulates are in the water - measured by how much light can pass through
Bioindicators (macroinvertebrate) - an organism that is sensitive to water quality.
Sedimentation- particles settling out of water.
Organic materials - material that decays that was once alive
Inorganic materials - materials that break down but were never alive like alluminum cans,
Vocabulary set 2 water cycle
First set of vocabulary
- Absorbency: The ability to take in a material.
- Capillary Action: The process that moves water through a narrow porous space.
- Adhesion: The tendency of water particles to stick to other surfaces.
- Cohesion: The tendency of water molecules to stick together. (Attractive force between water molecules)
- Surface Tension: The force that acts on the particles at the surface of a material to hold them together.
- Buoyancy: The ability of a fluid to exert an upward force on an object that is immersed in it.
- Polarity: Uneven sharing of electrons across a molecule.
- Universal Solvent: The quality of water that makes it able to dissolve more substances that any other solvent.
- Heat of Vaporization: The amount of heat required to change a substance from a liquid to a gas.
- Specific Heat: The amount of heat needed to raise the temperature of something 1 degree Celsius